Detection and quantification of novel C-terminal TDP-43 fragments in ALS-TDP
Abstract
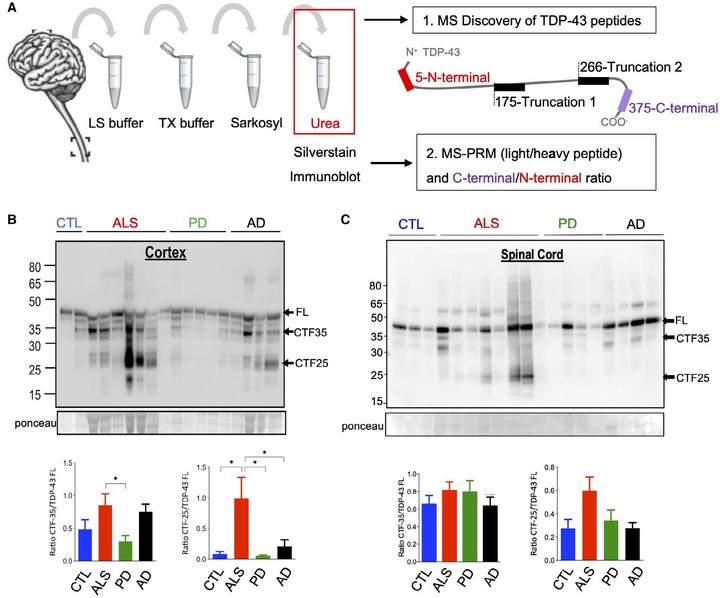
The pathological hallmark of amyotrophic lateral sclerosis (ALS) is the presence of cytoplasmic inclusions, containing C terminal fragments of the protein TDP‐43. Here, we tested the hypothesis that highly sensitive mass spectrometry with parallel reaction monitoring (MS‐PRM) can generate a high‐resolution map of pathological TDP‐43 peptide ratios to form the basis for quantitation of abnormal C‐terminal TDP‐43 fragment enrichment. Human cortex and spinal cord, microscopically staged for the presence of p‐TDP‐43, p‐tau, alpha‐synuclein, and beta‐amyloid pathology, were biochemically fractionated and analyzed by immunoblot and MS for the detection of full‐length and truncated (disease‐specific) TDP‐43 peptides. This informed the synthesis of heavy isotope‐labeled peptides for absolute quantification of TDP‐43 by MS‐PRM across 16 ALS, 8 Parkinson’s, 8 Alzheimer’s disease, and 8 aged control cases. We confirmed by immunoblot the previously described enrichment of pathological C‐terminal fragments in ALS‐TDP urea fractions. Subsequent MS analysis resolved specific TDP‐43 N‐ and C‐terminal peptides, including a novel N‐terminal truncation site‐specific peptide. Absolute quantification of peptides by MS‐PRM showed an increased C:N‐terminal TDP‐43 peptide ratio in ALS‐TDP brain compared to normal and disease controls. A C:N‐terminal ratio >1.5 discriminated ALS from controls with a sensitivity of 100% (CI 79.6–100) and specificity of 100% (CI 68–100), and from Parkinson’s and Alzheimer’s disease with a sensitivity of 93% (CI 70–100) and specificity of 100% (CI 68–100). N‐terminal truncation site‐specific peptides were increased in ALS in line with C‐terminal fragment enrichment, but were also found in a proportion of Alzheimer cases with normal C:N‐terminal ratio but coexistent limbic TDP‐43 neuropathological changes. In conclusion this is a novel, sensitive, and specific method to quantify the enrichment of pathological TDP‐43 fragments in human brain, which could form the basis for an antibody‐free assay. Our methodology has the potential to help clarify if specific pathological TDP‐43 peptide signatures are associated with primary or secondary TDP‐43 proteinopathies.